Heteranthera dubia
Metasp. Minnesota Valley, 138. 1892.
Plants perennial. Vegetative stems submersed and growing to surface, or emersed and contracted, procumbent. Flowering stems less than 5 mm, no visible distal internode. Sessile leaves alternate or appearing opposite at nodes with inflorescences, blade linear, thin; submersed leaves 3–10 cm × 1–5 mm, emersed leaves 2–4 cm × 1–3 mm. Petiolate leaves and stipules not produced. Inflorescences 1-flowered; spathes 12–45 mm, glabrous; peduncle absent. Flowers opening within 2 hours after dawn, wilting by dusk; perianth yellow, salverform, tube 1.5–11 cm, limb lobes equal, of uniform color, linear, 4–11 mm; stamens equal, 2–10 mm; filaments centrally inflated, glabrous; anthers coiling after anthesis, by early afternoon; style glabrous or scarcely pubescent. Seeds 7–30, 12–22-winged, 1–1.6 × 0.5–1.3 mm. 2n = 30.
Phenology: Flowering Apr–Nov.
Habitat: Streams, rivers, ponds, lakes
Elevation: 0–1500 m
Distribution
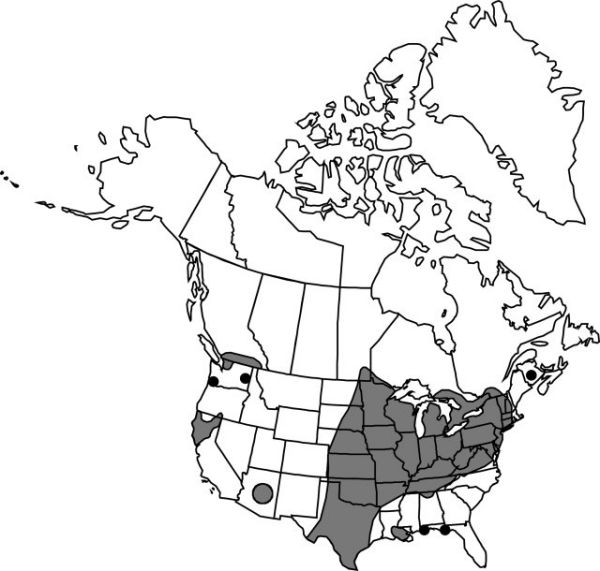
B.C., Man., N.B., Ont., Que., Ala., Ariz., Ark., Calif., Conn., Del., D.C., Fla., Ill., Ind., Iowa, Kans., Ky., La., Md., Mass., Mich., Minn., Mo., Nebr., N.H., N.J., N.Y., N.C., N.Dak., Ohio, Okla., Oreg., Pa., S.Dak., Tenn., Tex., Vt., Va., Wash., W.Va., Wis., Mexico, West Indies (Cuba).
Discussion
Extensive vegetative variation has been noted in the leaf morphology of Heteranthera dubia (C. N. Horn 1983). Plants in ponds and lakes tend to have longer, narrower leaves than plants from streams and rivers with a unidirectional current. Laboratory growth experiments revealed at least some genetic component to the differences (C. N. Horn 1981).
Plants are most extensive in their growth in alkaline waters (J. B. Moyle 1945), and, if plants are in a great enough density, they can cause an increase in water pH (E. B. Hollingsworth 1966). Growth in some lakes, streams, and canals has resulted in the species being considered a weed (E. B. Hollingsworth 1966).
Some authors (J. K. Small and J. J. Carter 1913; E. J. Alexander 1937) have transferred this species to the segregate genus Zosterella, based on the equal stamens (vs. nonequal in Heteranthera s.s.) and narrow, erect anthers (vs. broad and versatile). Small and Carter first described Zosterella citing only the above differences. Alexander later reported several more distinguishing characteristics: anthers coiling after anthesis, spathes sessile, and seeds relatively few. Recent molecular work by S. W. Graham et al. (1998) using chloroplast DNA suggests that Heteranthera dubia is clearly nested within the genus Heteranthera and is most closely related to H. seubertiana Solms, a tropical species.
Variation in perianth size has been a basis for separate recognition of Heteranthera liebmannii, or its equivalent, Zosterella longituba (E. J. Alexander 1937; D. S. Correll and H. B. Correll 1972). Alexander used differences in the lengths of perianth tube and lobes, and length of the perianth tube relative to that of the spathe to distinguish that segregate species. Study has revealed three causes of variation in perianth tube length: plants from populations farther north have a shorter perianth tube (herbarium study); flowers that develop under water have minimal perianth tube elongation, and the perianth remains completely within the spathe (J. W. Thieret 1972b; R. B. Wylie 1917); and lower overnight air temperatures result in less perianth tube elongation (controlled experiments). The specific epithet liebmannii was first applied to this entity by F. Buchneau (1872), under Potamogeton, and the type material consists of a series of specimens from Pital, Mexico, having only cleistogamous flowers. Later, the true generic identity of the specimens was realized. E. J. Alexander (1937), convinced that they represent a species distinct from those in Texas and Mexico, proposed the name Zosterella longituba for that entity, and placed Potamogeton liebmannii in synonomy. Since Alexander should have used the epithet liebmannii, which was not blocked in Zosterella, the name Z. longituba is illegitimate. An additional complication is that the types of P. liebmannii are morphologically ambiguous, since they include only cleistogamous flowers. Hence it is impossible, with current knowledge, to determine conclusively which of the two forms of Heteranthera dubia (as I recognize them) is represented by those types.
Populations are known to be infected by the fungal parasite Membranosorus heterantherae Ostenfeld & Peterson (H. S. Forest et al. 1986). The subsequent growths may be of two forms depending on whether they develop above the substrate on stems (nodal galls) or underground on roots (root galls).
Selected References
None.